|
The simplest aromatic hydrocarbon is the famous benzene molecule which consists of six carbon atoms and six hydrogen atoms as shown in figure 1. The six carbon atoms are bound into a closed, planar ring with the carbon atoms sitting on the vertices of a hexagon. Each carbon atom is further bound to a single hydrogen atom which lies exterior to the central ring. The aromatic character of the molecule, in practical terms, refers to the fact that the benzene molecule's ring-shaped structure makes its carbon-carbon skeleton more stable when compared to other types of hydrocarbon molecules.
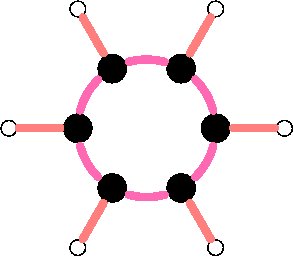
Figure 1: the stylized planar structure of a benzene molecule (C6H6). Carbon nuclei are indicated by black circles and hydrogen nuclei are indicated by white circles. The pink circular arcs represent the carbon-carbon aromatic bonds that form the "skeleton" of the molecule while the orange lines indicate the carbon-hydrogen covalent bonds that form the "decoration" around the exterior edge of the molecule.
The benzene ring structure has a unique fascination for astrochemists who study the cosmic complexity of carbon because it can be generalized in multiple ways to produce enormous classes of potentially interstellar molecules. One generalization of the benzene ring structure that is of particular interest to the Ames Astrophysics and Astrochemistry laboratory are the polycyclic aromatic hydrocarbons, also referred to as PAHs (monocylic benzene being a sort of honorary PAH). PAHs consist of multiple fused rings of carbon atoms (as in figure 2) with a benzene-like decoration of hydrogen atoms around their exterior perimeters. The cosmic complexity of PAHs arises from the exponential growth in the number of possible n-ring PAHs as a function of n. Considering that interstellar PAHs are estimated to have an average of roughly 50 carbon atoms, the number of potential PAH structures is very large indeed.
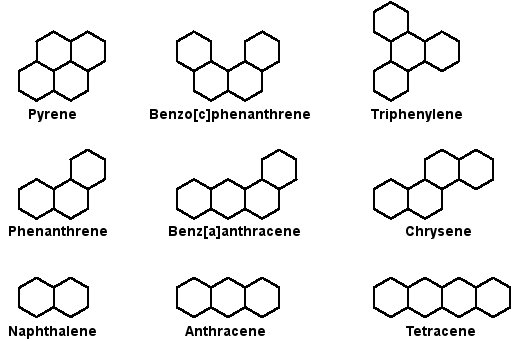
Figure 2: the highly stylized planar structures of some simple, benzenoid PAHs
Polycyclic aromatic hydrocarbons with a "decoration" that consists only of hydrogen atoms are commonly represented by simple hexagons. The vertices of the hexagons indicate the positions of the carbon atoms and the edges of the hexagons indicate the aromatic "skeleton" of the molecule. Hydrogen atoms bound to each second-order vertex (i.e. a vertex that borders only a single ring) are assumed. Shown here are the aromatic skeletons of PAHs with two to four rings.
The PAHs are plausible candidates for an interstellar molecule for a number of reasons. First, PAHs are very easily formed in hot (T ~ 2000 K) carbon and hydrogen rich gases. On Earth, these conditions are achieved in combustion processes such as burning wood or burning gasoline (thus making wood smoke and automobile exhaust major sources of PAH pollution). In the interstellar medium, gaseous outflow from carbon-rich red giants is a major source of simple carbon-bearing molecules such as carbon monoxide and acetylene (C2H2, or if your prefer, "one-third" of a benzene ring).
On Earth and in the interstellar medium, a high temperature makes it easy for a hydrocarbon to lose one or more hydrogen atoms. A high temperature and a high gas density also promote frequent collisions between hydrocarbon molecules. Hydrocarbon molecules with missing hydrogen atoms are more reactive and therefore more likely to capture carbon atoms in these collisions. The net effect is that the chemistry of the hot gas mixture favors growth from small hydrocarbons to larger ones with PAHs commonly being one of the end products.
Second, PAHs are also generally highly efficient at processing the interstellar UV radiation field into infrared radiation. When a PAH molecule absorbs a UV photon, this energy input can be extremely rapidly distributed throughout the entire molecule. The effect of this energy distribution is to cause the molecule's constituent atoms to begin vibrating relative to each other, thus producing distortions in the overall structure of the molecule. After a UV excitation, a vibrating PAH molecule will very rapidly begin to relax back to its unexcited, non-vibrating state by converting this vibrational energy into emitted infrared energy. The net effect of this process of excitation and relaxation is that the original UV photon has been converted into a much larger number of infrared photons. PAHs should therefore be especially conspicuous in interstellar environments through their IR emission.
In fact, exactly this type of interstellar emission has been observed in the form of the so-called "unidentified infrared bands", now commonly referred to as the "aromatic infrared bands" (see figure 4). These emission features are characterized by dominant features near 3030, 1610, 1280, 1150, and 890 cm-1 (3.3, 6.2, 7.7, 8.6, and 11.2 microns respectively) as well as a number of more minor bands and underlying continua. The brightest emission is observed from dusty regions exposed to intense ultraviolet radiation. Similar emission spectra are observed from objects which span the lifecycle of matter in the interstellar medium (ISM) -- from objects only a few thousand years in age (late carbon star ejecta and planetary nebulae shells) to those millions of years in age (HII regions, reflection nebulae, and the diffuse ISM).
The attribution of these bands to PAHs is based on several pieces of circumstantial evidence which point to these species. First, the banded (rather than continuous) nature of the spectrum, the typically large feature/continuum ratio, and the close association of this emission with ultraviolet radiation all indicate that the emission is due to infrared fluorescence from gas phase molecules excited by the absorption of a single ultraviolet or visible photon rather than thermal emission from a solid material. Second, careful observations of planetary nebulae have established that the fraction of the total infrared energy that is emitted through these features is closely correlated with the amount of available carbon, indicating that the gas-phase molecular carriers are carbon-rich. Third, since the carbon-rich carriers must survive under remarkably harsh conditions, they must also be extremely stable. Finally, although there are variations among the relative band intensities, the features are correlated, implying that a single class of chemical species is responsible for these spectral signatures. The cosmic complexity of the PAHs readily accommodates all of these constraints.
|